 It's likely that the exact dependency is very complicated and nowadays, such atomic properties are mostly calculated computationally anyway, which removes the need for such a mathematical formula. The description of $Z_\mathrm$ (going from 1.28 to 2.51), although it's sadly difficult to find an exact mathematical formulation in any textbook. So what am I missing here? Why does shielding matter? Is the force on a single valence electron a function of the net nuclear charge or do we have to look at it from a more piecewise level, considering how other electrons are in between it and the nucleus and how it interacts with those individual electrons? 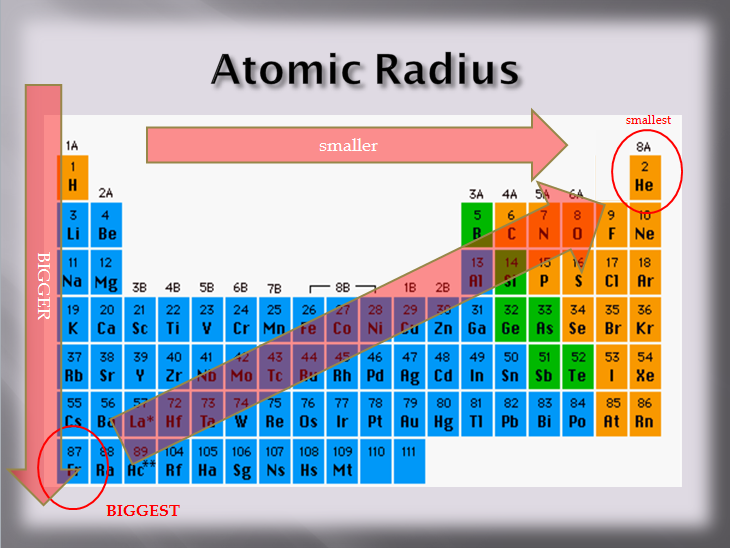 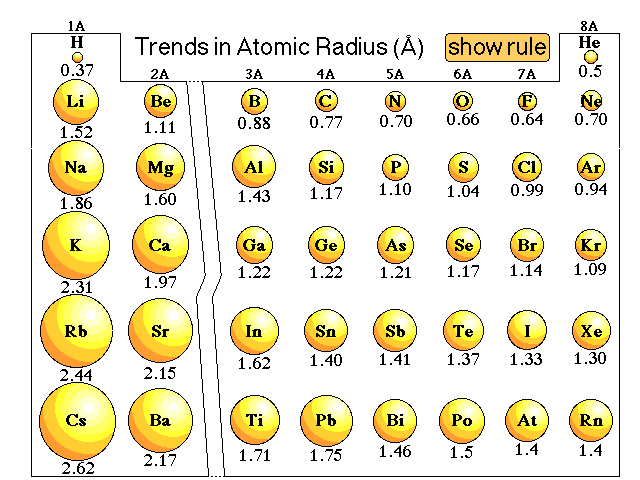
I find this contradictory, since as we go down a group, the effective nuclear charge remains constant (equal numbers of protons and non-valence electrons are added). Thus with more energy levels present and more shielding, valence electrons begin to reside further and further away from the nucleus.īut at the same time, textbooks make a big deal about the concept of effective nuclear charge, which states that the "effective" force a given valence electron feels is a function of the net charge of the nucleus and non-valence electrons combined-the effective nuclear charge. The general explanation seems to state that as we go down a group, there are more electron shells present to contribute to a "shielding" phenomenon, where inner electrons cancel out part of the inward force from the nucleus with a repulsive force. I realize this should be a fairly basic question, but I'm still not quite satisfied with what I've been told from numerous sources. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
